Breathe!
No one knows of any other planet where you can do this simple thing.
Other planets and moons in our solar system have atmospheres, but none of them could support life as we know it. They are either too dense (as on Venus) or not dense enough (as on Mars), and none of them have much oxygen, the precious gas that we Earth animals need every minute.
So how did our atmosphere get to be so special?
Some scientists describe three stages in the evolution of Earth’s atmosphere as it is today.
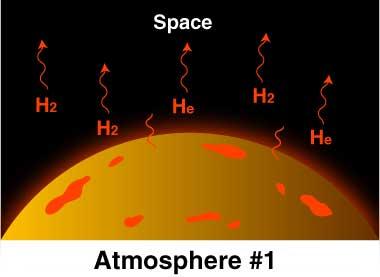
Just formed Earth: Like Earth, the hydrogen (H2) and helium (He) were very warm. These molecules of gas moved so fast they escaped Earth's gravity and eventually all drifted off into space.
Earth’s original atmosphere was probably just hydrogen and helium, because these were the main gases in the dusty, gassy disk around the Sun from which the planets formed. The Earth and its atmosphere were very hot. Molecules of hydrogen and helium move really fast, especially when warm. Actually, they moved so fast they eventually all escaped Earth's gravity and drifted off into space.
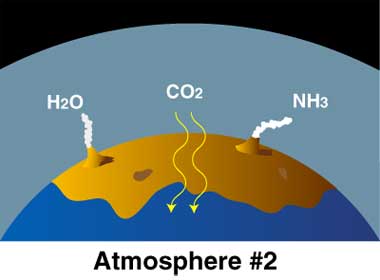
Young Earth: Volcanoes released gases H2O (water) as steam, carbon dixoide (CO2), and ammonia (NH3). Carbon dioxide dissolved in seawater. Simple bacteria thrived on sunlight and CO2. By-product is oxygen (O2).
Earth’s “second atmosphere” came from Earth itself. There were lots of volcanoes, many more than today, because Earth’s crust was still forming. The volcanoes released
- steam (H2O, with two hydrogen atoms and one oxygen atom),
- carbon dioxide (CO2, with one carbon atoms and two oxygen atoms),
- ammonia (NH3, with one nitrogen atom and three hydrogen atoms).
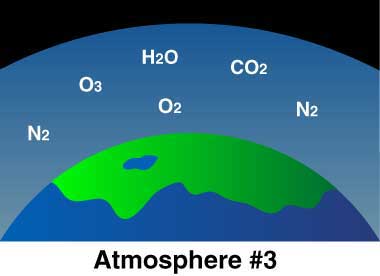
Current Earth: Plants and animals thrive in balance. Plants take in carbon dioxide (CO2) and give off oxygen (O2). Animals take in oxygen (O2) and give off CO2. Burning stuff also gives off CO2.
-
Much of the CO2 dissolved into the oceans. Eventually, a simple form of bacteria developed that could live on energy from the Sun and carbon dioxide in the water, producing oxygen as a waste product. Thus, oxygen began to build up in the atmosphere, while the carbon dioxide levels continued to drop. Meanwhile, the ammonia molecules in the atmosphere were broken apart by sunlight, leaving nitrogen and hydrogen. The hydrogen, being the lightest element, rose to the top of the atmosphere and much of it eventually drifted off into space.
Now we have Earth’s “third atmosphere,” the one we all know and love—an atmosphere containing enough oxygen for animals, including ourselves, to evolve.
So plants and some bacteria use carbon dioxide and give off oxygen, and animals use oxygen and give off carbon-dioxide—how convenient! The atmosphere upon which life depends was created by life itself.




